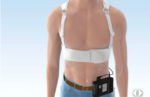
A federal judge has denied Asahi Kasei (TYO:3407) subsidiary Zoll Medical’s motion to toss out a lawsuit filed by the family of a woman who attribute her death to a faulty Zoll LifeVest wearable defibrillator.
U.S. District Court Judge Darrin Gayles in the Southern District of Florida also halted Zoll’s effort to bar evidence from the plaintiffs’ expert witness, an engineering professor who has taught courses on soldering and resoldering.
Zoll and the family of Debra Godelia agreed that the LifeVest that she was wearing in November 2013 failed to deliver a shock during a defibrillation event. Godelia became unconscious and died in the hospital two days later.
The family claims that defective soldering caused the cable connecting the rear therapy electrode set to the LifeVest’s distribution network to separate from the network’s circuit board. Zoll contends that the separation must have taken place after the event.
The family also claims that Zoll failed to implement processes to ensure that its manufacturing of the LifeVest, and in particular, the soldering at issue, was non-defective and that the alleged soldering defect was a direct result of Zoll’s failure to implement such manufacturing processes. Zoll disagrees.
In his order, Gayles said that a jury would need to decide whether the family’s claims are correct.
“While there might not have been any other appropriate external or internal defibrillation device for Mrs. Godelia, she might have chosen, as an alternative option, to not wear the defibrillation device at all and seek another form of treatment,” Gayles said. “Without the LifeVest, her family, who had been instructed by Defendant to stay away from Mrs. Godelia while the LifeVest sounded an alarm, might have attempted CPR the minute she fell to the ground. As with the questions surrounding the detached cable, questions about Mrs. Godelia’s reliance on Defendant’s representations are for a jury.”
Zoll did not immediately respond to a request for comment on the judge’s orders.

