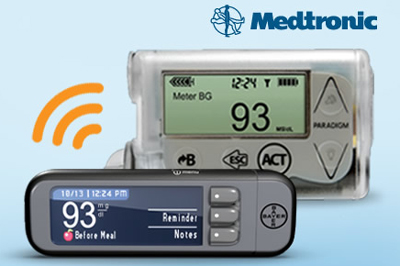
Bayer (PINK:BAYRY) today announced that it won FDA clearance to pair its Contour Next Link blood glucose meter with Medtronic’s (NYSE:MDT) newly approved MiniMed 530G with Enlite, the 1st "artificial pancreas" system for patients with diabetes.
Bayer’s meter is the 1st device with the FDA’s OK to pair with the new MiniMed system, the 1st U.S.-approved device of its kind to feature threshold suspend automation, the ability to automatically stop halt glucose delivery at a preset level. The Contour Next Link measures blood glucose levels to regulate insulin delivery by MiniMed 530G.
Bayer’s system can also wirelessly transmit blood glucose test results to Medtronic’s insulin devices and communicate via Medtronic’s CareLink software, according to a press release.
Medtronic late last month announced that it had won FDA approval for the MiniMed 530G with Enlite, labeled for diabetics aged 16 and older. Medtronic and other medical device makers have been racing to develop the 1st true artificial pancreas systems, considered "the Holy Grail" of diabetes research. The Juvenile Diabetes Research Foundation and Becton Dickinson & Co. (NYSE:BDX) recently said they’re "accelerating" development of the technology by combining their proprietary diabetes management devices in a 3-year agreement. And Cambridge Consultants and the U.K.’s Institute of Metabolic Science said they’re also researching artificial pancreas technology at Addenbrooke’s Hospital in Cambridge, England.
The MiniMed 530G features "threshold suspension," a program which monitors and modifies insulin delivery when blood glucose readings hit preset levels, helping reduce the incidence of hypoglycemia without affecting blood sugar. In a clinical trial of nearly 250 patients with Type I diabetes and nocturnal hypoglycemia, researchers reported that acute events were 31.8% less likely in patients using the device with threshold suspension. Medtronic said it plans to begin ramping up production immediately ahead of a planned launch "in the next several weeks."

