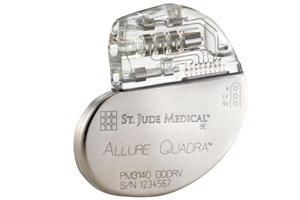
European regulators granted market approval to St. Jude Medical (NYSE:STJ) for its Allure Quadra quadripolar cardiac resynchronization therapy pacemaker, the medical device giant announced this week.
The Allure Quadra is the 1st device of its kind on the European market, leveraging quadripolar lead technology to offer more varied pacing options.
The news didn’t seem to stir up much enthusiasm from Wall Street, where STJ shares were down 1.1% to $41.22 as of about 12:15 p.m. today.
The Allure Quadra is the latest in a string of St. Jude quadripolar cardiac rhythm management implants to hit markets around the world in recent years. In July 2012 the medical device company launched its Unify Quadra CRT defibrillator onto the Japanese market. That device hit markets in Europe in May 2010, launched in India in September 2011, won FDA approval in November 2011.
"Since its launch in 2010, the St. Jude Medical quadripolar CRT-D platform has allowed physicians to more effectively treat their patients and we expect the same results with our quadripolar CRT-P line," implantable electronics systems division president Dr. Eric Fain said in prepared remarks. "We believe these benefits result in a cost-effective solution that elevates the standard of care for heart failure patients in need of cardiac resynchronization therapy."
The new CRM platform features the only CE Mark-approved stroke risk identification algorithm, according to St. Jude, and the Allure pacemaker has access to diagnostic data, the CorVue impedance monitoring system and Direct Trend device data reports.

