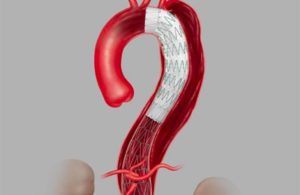
Cook Medical has launched its Zenith dissection endovascular system, recently approved by the FDA.
The first patient was treated with the system at Cooper University Health Care in Camden, N.J., Cook Medical announced yesterday.
The Zenith system was designed to provide physicians with a less-invasive alternative to open surgery for repair of Type B dissections of the descending thoracic aorta. Zenith consists of a proximal stent-graft component and a distal bare-stent component.
Get the full story on our sister site, Medical Design & Outsourcing.

