St. Jude Medical pushes 'play' on Portico TAVI device
March 12, 2015 by Brad Perriello
St. Jude Medical, which paused implantations of its Portico replacement heart valve last fall out of an abundance of caution, told MassDevice.com today that European authorities reinstated Portico’s CE Mark.
The decision means St. Jude is resuming clinical and commercial implants of the Portico 23mm and 25mm transcatheter aortic valve implants in Europe, spokesman Justin Paquette told us via email. Read more
![]()
![]()
FDA clears Tornier's Simpliciti shoulder repair device
March 10, 2015 by Brad Perriello
Tornier today said the FDA cleared its new Simpliciti shoulder repair device for patients whose osteoarthritis or traumatic arthritis resulted in severely painful and/or disabled shoulder joints.
Amsterdam-based Tornier, which is gearing up for a $3.3 billion merger-of-equals with Wright Medical, said the safety watchdog based its 510(k) clearance on a 3-year investigational device exemption trial of the Simpliciti device. Read more
![]()
Japan approves Lombard Medical's AorFlex delivery device
March 13, 2015 by Brad Perriello
Lombard Medical Technologies today said it won approval from the Japanese Ministry of Health for the AorFlex system used to implant its Aorfix stent graft, which is used to repair abdominal aortic aneurysms.
The Aorfix device won approval from the Japanese Ministry of Health last August, making it the 1st endovascular stent graft approved in Japan to treat aortic neck angulations up to 90°. Read more
![]()
FDA issues new guidance on scopes linked to deadly 'superbug' infections
March 12, 2015 by Brad Perriello
The FDA today issued new guidance for makers of reusable medical devices like the endoscopes linked to a rash of deadly "superbug" infections.
The new guidance, “Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling,” is aimed at helping medtech makers develop safer devices, the federal safety watchdog said. Read more
![]()
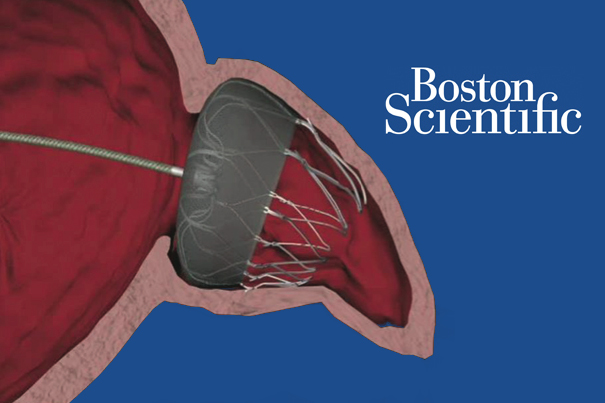
Boston Scientific jumps on Watchman approval
March 16, 2015 by Brad Perriello
Investors sent shares of Boston Scientific up nearly 4% today after the medical device company said the FDA approved its long-delayed Watchman anti-stroke device.
BSX shares were trading at $17.20 apiece in mid-day activity today, up 3.6%, as investors reacted to the decade-long odyssey Boston Scientific undertook in getting Watchman on the U.S. market. Read more