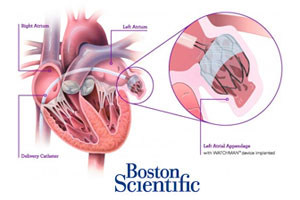
The Watchman heart implant made by Boston Scientific (NYSE:BSX) improved quality-of-life measures more than warfarin in atrial fibrillation patients after 1 year, according to a study in the Journal of the American College of Cardiology.
The more than 700-patient Protect AF trial produced QOL scores that were ahead of warfarin for the study’s primary efficacy endpoint (stroke, cardiovascular death or systemic embolism after 1,000 patient-years) in patients treated with the Watchman left atrial appendage closure device, according to heartwire.
But the risk of major bleeding, cardiac perfusion and device embolization (the trial’s primary safety endpoint) was higher in Watchman patients, according to the study.
"The improvements were mainly in the area of physical functioning and were seen in both warfarin naive and not-warfarin-naive patients treated with LAA closure," wrote the authors, led by Dr. Oluseun Alli of the University of Alabama.
Natick, Mass.-based Boston Scientific acquired the Watchman device along with AtriTech for up to $375 million in 2011. Last summer the company closed enrollment in a 2nd trial of the device, with more than 2,000 subjects signing on for the randomized comparison of Watchman with warfarin in high-risk patients with atrial fibrillation. In August the device won expanded CE Mark approval in the European Union for the catheter-based clot-capture system, to patients with atrial fibrillation who can’t be treated with warfarin or new oral anticoagulants.

