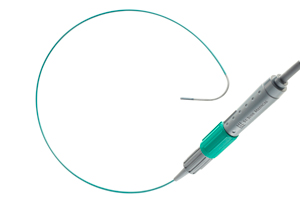
Medical device maker St. Jude Medical (NYSE:STJ) won CE Mark approval in the European Union for its ViewFlex Xtra intracardiac echocardiography catheter for use with the device giant’s ViewMate Z ultrasound console.
The upgraded system offers one-handed use, an ergonomic handle and 4-way tip deflection, according to a press release.
The ViewFlex Xtra Ice, which won FDA 510(k) clearance in July 2012, helps physicians generate high-resolution images of a patient’s blood flow and cardiac anatomy in real time.
The device is an adjunct to St. Jude’s ViewMate Z combination echocardiography and convertible ultrasound system, developed in collaboration with Mountain View, Calif.-based Zonare Medical Systems.
"The combination of the ViewFlex Xtra ICE Catheter and ViewMate Z Intracardiac Ultrasound Console in an integrated EP Lab not only yields exceptional images, but more importantly allows physicians to focus on the patient and less on catheter manipulation," St. Jude cardiovascular & ablation technologies president Frank Callaghan said in prepared remarks.
STJ shares got a boost today, rising 2% to trade at $39.32 as of about 11:45 a.m.

