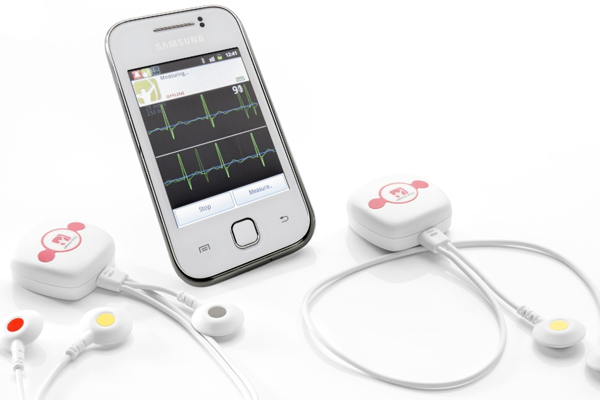
Finnish medical device maker Mega Electronics won FDA approval for its eMotion continuous ECG monitor, designed to work with Android-based smartphones.
The system provides real-time remote heart rhythm monitoring that collects data and provides caregivers with alarms when abnormal heart rhythms are detected. The device is comprised of wireless electrodes that communicate via Bluetooth with a smartphone app.
Mega Electronics will have to face rival AliveCor and its smartphone ECG monitor, available on iPhones and, as of October, on Android phones. Unlike the eMotion system, AliveCor’s device is not designed for continuous monitoring. Both devices must be prescribed by a physician.
The eMotion system features a few additional perks, including a GPS system that can alert emergency services of a patient’s location in case of emergency and software and the flexibility to incorporate Bluetooth-based blood pressure and weight monitoring, according to company’s website.

