Medical device maker Medtronic (NYSE:MDT) this week touted FDA clearance for its next-generation FlexCath Advance steerable sheath, launching U.S. commercial efforts immediately.
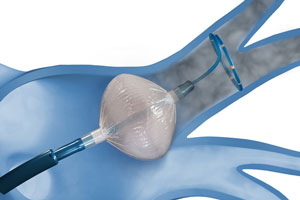
The new FlexCath Advance sheath is designed to work with Medtronic’s Arctic Front Advance cryoablation balloon system, which treats irregular heartbeat by blocking electrical signals that trigger erratic rhythms in the heart’s upper chambers.
The new sheath has several design improvements and is easier to use, according to a press release.
"Building upon the Arctic Front System’s proven platform, the FlexCath Advance Steerable Sheath augments the overall efficiency of the procedure," Medtronic vice president and AF Solutions general manager Reggie Groves said in prepared remarks. "It completes the Arctic Front Advance System and is the 4th new product we have launched in the U.S. market in 2 years."
Arctic Front Advance and an earlier generation of the device have treated more than 50,000 in 32 countries, according to Medtronic.
The Minnesota-based medtech giant in August 2012 announced U.S. and European approvals for the Arctic Front Advance system, predicting rapid adoption among clinicians, given the success of its predecessor.
Medtronic in January 2012 enrolled the 1st patient in its Fire & Ice ablation study, comparing its 1st generation Arctic Front system to Biosense Webster’s Carto system-guided Thermocool catheter.
The Fire & Ice study, expected to enroll up to 572 patients, is the largest study to date comparing 2 atrial fibrillation ablation systems.

