
Say hello to MassDevice +7, a bite-sized view of the top seven med-tech stories of the week. This latest feature of MassDevice.com’s coverage highlights our seven biggest and most influential stories from the week’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
If you read nothing else this weekend, make sure you’re still in the know with MassDevice +7.
7. Consumer group demands more med-tech oversight from MDUFMA III

Consumer Union demanded that a new user fee deal the medical device industry brokered with the FDA include more stringent oversight of med-tech makers. "We shouldn’t be used as guinea pigs when it comes to something as serious as a medical implant," according to the organization’s website. "Medical devices should not be ticking time bombs!"
![]()
6. MassDevice Podcast: Soteira CEO Larry Jasinski

Soteira CEO Larry Jasinski is no stranger to adversity. In the last 5 years the spinal fusion device maker has overcome criticism from the research community and mid-application surprises from the FDA, but its newest 510(k) win is a testament to its persistence.
![]()
5. Smith & Nephew pays $22M fine for 10 years of bribery in Greece

British orthopedic titan Smith & Nephew (NYSE:SNN) must pay $22.2 million to settle charges of bribing Greek doctors for more than a decade, according to SEC documents. The complaint alleges that Smith & Nephew subsidiaries used a distributor that created a "slush fund" to bribe doctors working at government hospitals in Greece.
![]()
4. AdvaMed: Repealing the med-tech levy is about tax reform, not health care reform

AdvaMed officials released a set of principles for modifying the U.S. tax system, part of the group’s efforts to gain Democratic support for repealing the impending 2.3% medical device tax by separating the issue from health care reform.
![]()
3. Medicare proposes TAVI reimbursement guidelines
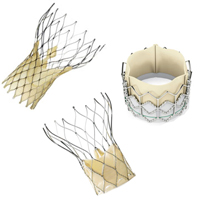
The Centers for Medicare & Medicaid Services released a proposal detailing guidelines for reimbursement of transcatheter aortic valve implantation procedures for treating severe symptomatic aortic stenosis.
![]()
2. Boston Scientific to hit the acquisitions trail
Boston Scientific (NYSE:BSX) plans to hoard most of the more than $1 billion in free cash flow it expects to generate this year so it can make acquisitions to boost its top line, CFO Jeff Capello told analysts.
![]()
1. Stryker CEO Stephen MacMillan resigns for “family reasons”

Stryker Corp. (NYSE:SYK) chairman, president & CEO Stephen MacMillan walked away from one of the most successful medical device companies in the world on Wednesday, citing family reasons for an abrupt departure.

