Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
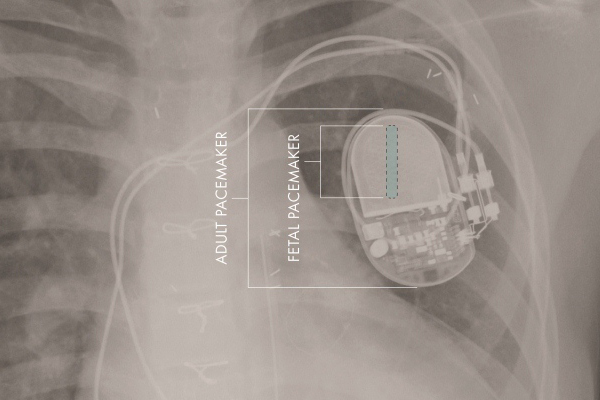
3. FDA grants exemption for fetal micro-pacemaker
Researchers at Children’s Hospital Los Angeles and the University of Southern California said the FDA granted a humanitarian device exemption for the fetal micro-pacemaker they designed.
The device is the 1st fully implantable pacemaker for use in a fetus with complete heart block, according to a press release. The 1st implantation in a human is expected “in the near future,” the researchers said. Read more
![]()
![]()
2. Stryker debuts Osiris bone putty
More than 2 months after Osiris Therapeutics struck an exclusive marketing deal with Stryker for its Bio4 bone putty, the agreement is starting to bear fruit.
Stryker this week launched the Bio4 bone allograft during the American Academy of Orthopaedic Surgeons annual meeting in Las Vegas. Read more
![]()
1. European regulators OK $13B Zimmer-Biomet deal
Zimmer said that anti-trust regulators in the European Union approved its pending merger with cross-town orthopedics rival Biomet, setting the stage for the $13.35 billion deal to close next month.
Warsaw, Ind.-based Zimmer said the European Commission cleared the deal on a conditional basis, provided the company goes through with its plan to divest certain assets. Read more