
Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
3. Medtronic wins FDA approval for more Attain Performa quad defib leads
Medtronic said today that it won pre-market approval from the FDA for another pair of its Attain Performa defibrillator leads, designed for varying vessel sizes and curvatures.
Medtronic said the Attain Performa S-shaped and straight leads are meant for its Viva Quad XT and Viva Quad S CRT-Ds. Read more
![]()
![]()
2. Study buoys Medtronic, Edwards valves in bicuspid disease
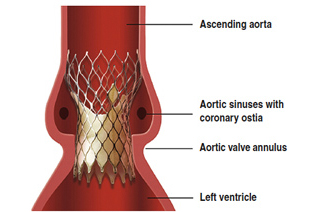
Medtronic and Edwards Lifesciences are fierce and sometimes bitter rivals in the field of transcatheter aortic valve implantation, but both companies’ implants scored in a clinical study that looked at their use for a specific condition.
Researchers from Europe, Canada and Israel determined that TAVI devices produced encouraging results in a small group of patients with bicuspid aortic valve disease. The findings are detailed in the latest edition of the Journal of the American College of Cardiology. Read more
![]()
1. St. Jude Medical wins CE Mark for Quadra Allure MP pacemaker
St. Jude Medical said today that it won CE Mark approval in the European Union for its Quadra Allure MP cardiac resynchronization therapy pacemaker, touting it as the world’s 1st to offer multi-point pacing.
The technology allows physicians to pace multiple locations on the left side of the heart to optimize CRT pacing using St. Jude’s 4-electrode Quartet LV lead, according to a press release. Read more



