Say hello to MassDevice +3, a bite-sized view of the top three medtech stories of the day. This feature of MassDevice.com’s coverage highlights our 3 biggest and most influential stories from the day’s news to make sure you’re up to date on the headlines that continue to shape the medical device industry.
3. Medtronic and Covidien reveal their integration teams
This week merging device makers Covidien and Medtronic jointly named more than 45 people to take an intimate interest in the companies’ high-profile integration.
The appointees include employees from both sides who have been tasked with coordinating the finer points of the $43 billion mega-merger. The teams will tackle all specific functions and business areas under the supervision of the Joint Steering Committee, which is comprised of senior leaders from both Medtronic and Covidien. Read more
![]()
![]()
2. Medtronic claims a 40% share of U.S. TAVI market with CoreValve
Medtronic executives yesterday said the company captured more than 40% of the U.S. market for transcatheter aortic heart implants from Edwards Lifesciences in just 2 quarters.
CoreValve was 1st to market with a TAVI device when it won CE Mark approval in the European Union in 2007. Read more
![]()
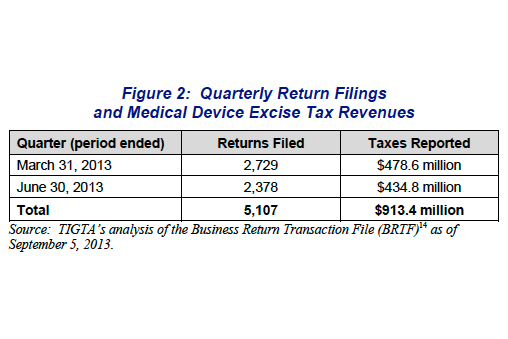
1. Report: The IRS is having trouble with the medical device tax
Not even the IRS can make sense of the medical device tax, a provision of the Affordable Care Act requiring medical device companies to pay a 2.3% levy on all U.S. sales, according to a report from a U.S. Treasury inspector general.
The tax is imposed on all U.S. sales of prescribed medical devices and contains a “retail exemption” on devices sold to the general public, such as contact lenses or diabetes devices and supplies. Estimates on the amount the tax is expected to range vary, from $30 billion over 10 years to the Joint Committee on Taxation’s forecast for $20 billion for fiscal years 2013 through 2019. Read more