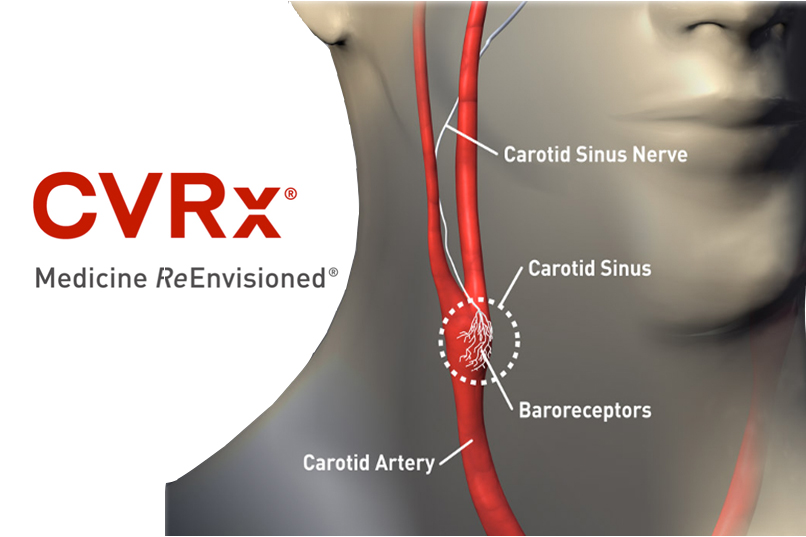
The principal investigator in an early feasibility study of CVRx‘s Barostim device said today that the company won FDA approval for a pivotal trial later this year, after reporting strong results from an early feasibility trial of the device.
"The results of this study are remarkable," Dr. Michael Zile of the Medical University of South Carolina said today at the annual Heart Rhythm Conference in Boston. "This represents a therapy which can be applied to those patients in which you have exhausted all other therapies and it can be applied to those patients who are not candidates for CRT."
The 140-patient Phase II trial enrolled heart failure patients treated with optimal drug regimens. Subjects were randomized 1:1 to receive the Barostim device plus optimal treatment; about ⅓ had already been implanted with cardiac resynchronization therapy devices, Zile said.
"We found that the response to [baroreflex activation therapy] seemed more pronounced in patients with no CRT than with CRT," he said. "Obviously all of these results have to be confirmed in a large prospective randomized clinical trial."
Zile said CVRx has planned and won approval for a randomized pivotal trial from the FDA, planned for September of this year, that’s slated to enroll 480 patients in 90 centers. The goal for the trial, which will not enroll patients with CRT devices, is to generate data on 320 outcomes.