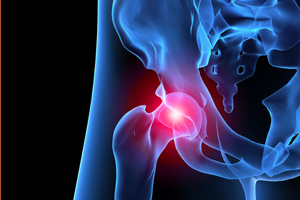
Wright Medical (NSDQ:WMGI) unveiled in its latest earnings report that the medical device company received a subpoena from the U.S. Attorney’s Office for the Western District of Tennesee requesting records relating to the its suite of metal hip implants.
The office requested documentation covering the period from Jan. 1, 2000, to Aug. 2, 2012, and Wright Medical is in the process of gathering the required data, according to the report.
Wright’s Profemur line, which includes metal hip stems and total hip replacement systems, may also be at risk following Stryker’s (NYSE:SYK) recall of a competing device, according to the company.
Last month Stryker recalled a pair of hip implants and halted global production over concerns that the devices may be prone to "fretting and/or corrosion at or about the modular-neck junction," which may lead to pain, swelling and adverse reactions in surrounding tissue.
"Although Stryker’s recalled modular hip stems differ in design and material from our Profemur modular neck hip stems, there is a risk that Stryker’s recall and the resultant publicity could negatively impact sales of modular neck systems of other manufacturers, including our Profemur system, even if the issues cited by Stryker are unique to Stryker products," according to Wright’s report.
Wright Medical did not immediately return requests for comment.

