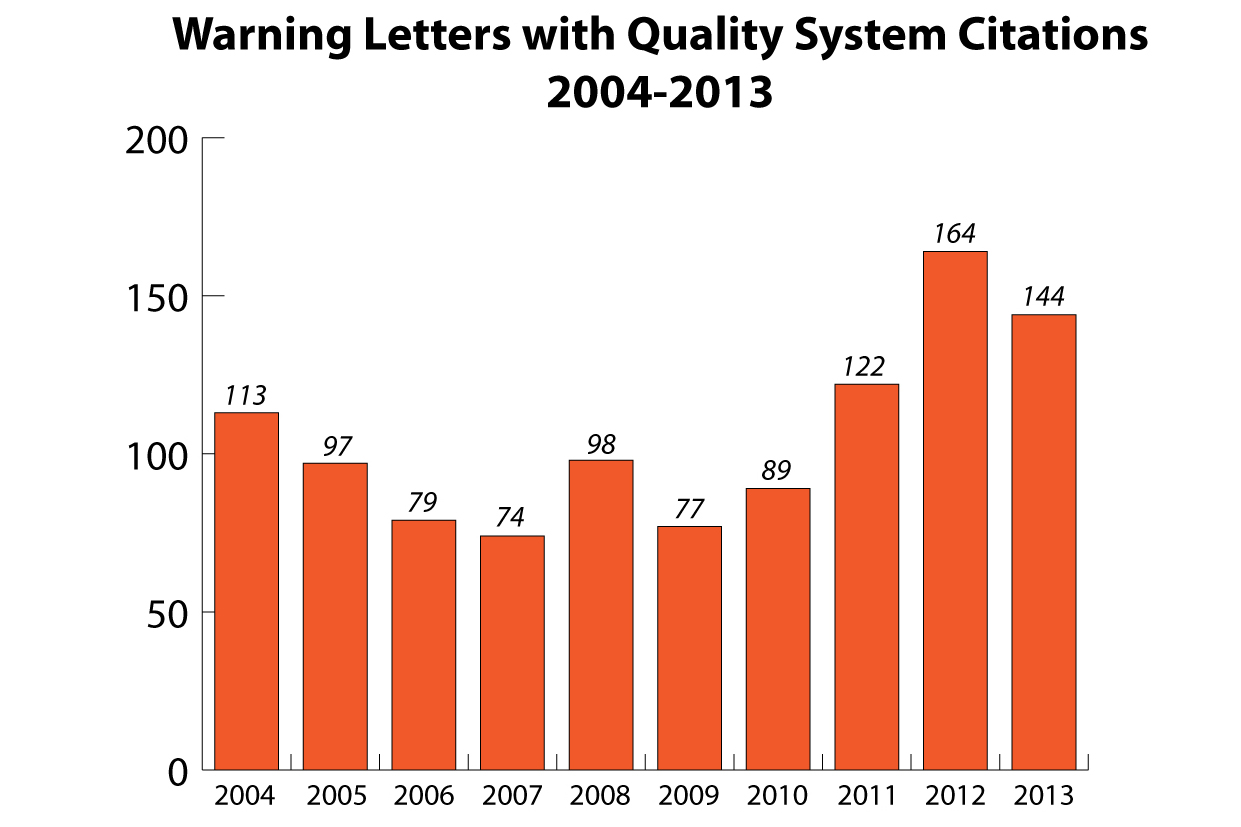
The number of warning letters sent by the FDA to medical device companies dipped for the 1st time since 2009, according to a report from the agency’s Center for Devices & Radiological Health.
The CDRH sent 144 warning letters to medtech makers in 2013, according to the report, compared with 164 last year, a 12.2% decline, according to the report. Of the U.S. firms inspected by the agency, 4% received warning letters, the agency said.
Sixteen percent of foreign firms inspected were flagged by the FDA, led by Germany with 86 site visits from agency personnel, followed by China (82), Canada (35), France (33) and Japan (24), according to the report.
Although there were fewer warning letters issued in 2013 compared with 2012, the number of overall inspectional observations rose 17%, according to the report.