Boston Scientific (NYSE:BSX) CFO Dan Brennan said today that the FDA wants to hold another advisory panel hearing on its Watchman anti-stroke device and pushed the approval timeline for the Watchman heart implant into next year. Brennan, speaking today at the Wells Fargo Healthcare Conference, said Boston Scientific is now hoping for an approval during the 1st half of 2015.
The as-yet-unscheduled panel would be the 3rd advisory committee hearing over Boston Scientific’s pre-market approval application for the Watchman device and comes despite positive votes from the 2 prior meetings.
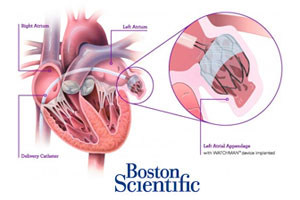
Despite the 1st panel’s 2009 approval recommendation, the FDA in March 2010 issued a "not approvable" letter for the Watchman device, a transcatheter implant designed to reduce the risk of stroke by closing off the heart’s left atrial appendage. A 2nd panel voted 13-1 last year that the Watchman implant meets safety and efficacy goals.
Today Brennan said the federal watchdog agency wants a 3rd hearing to consider "the totality of the clinical evidence" gathered so far.
Brennan was mum when pressed by Wells Fargo analyst Larry Biegelson for specifics on the FDA’s concerns.
"We can’t get into the specifics of the individual conversations with the FDA," he said. "Their desire is to look at the total body of clinical evidence up to the present time. We look forward to presenting the data and believe that the data and what we have support the safety and efficacy of Watchman in appropriately selected patients.
BSX shares were down 2.9% to $12.54 apiece as of about 1 p.m. Eastern today.

