
FDA wants another panel hearing on Boston Scientific's Watchman anti-stroke device
June 17, 2014 by Brad Perriello
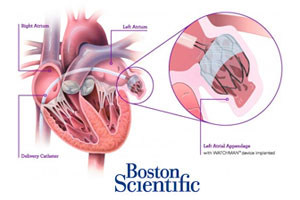
Boston Scientific CFO Dan Brennan said today that the FDA wants to hold another advisory panel hearing on its Watchman anti-stroke device and pushed the approval timeline for the Watchman heart implant into next year. Brennan, speaking today at the Wells Fargo Healthcare Conference, said Boston Scientific is now hoping for an approval during the 1st half of 2015.
The as-yet-unscheduled panel would be the 3rd advisory committee hearing over Boston Scientific’s pre-market approval application for the Watchman device and comes despite positive votes from the 2 prior meetings. Read more
![]()
![]()
FDA talks Twitter and other online etiquette for medtech makers
June 19, 2014 by Arezu Sarvestani

FDA regulators published a pair of new draft guidances this week to provide clarity on its stance regarding online communication in challenging situations, especially via character-constrained channels such as Twitter or when responding to misinformation posted online.
The 1st guidance offers recommendations on communicating through mediums that have character space limits, referencing Twitter’s 140-character Tweets and paid search links offered by web browsers, but also attempting to look to future channels. The guidance doesn’t impose any enforceable standards, but reflects “the Agency’s current thinking on a topic.” Read more
![]()
Malaysian medical device market regulators clarify, expand policies
June 20, 2014 by MassDevice

Medical device market regulators in Malaysia have implemented several new policiespertaining to the country’s recently enacted Medical Device Act.
Recent Medical Device Authority (MDA) policies target five key areas: authorized representation, Conformity Assessments, registration exemptions, export/import requirements and Good Manufacturing Practice (GMP) certifications for establishment licensing. Read more
![]()
FDA issues draft guidances for industry on social media and internet communications about medical products: Designed with patients in mind
June 17, 2014 by MassDevice

Ongoing changes in technology transform medical products – and the ways that both patients and health care providers learn about those products. In today’s world, in addition to traditional sources of medical product information, patients and health care providers regularly get information about FDA-regulated medical products through social media and other Internet sources, and those technologies continue to evolve.
But regardless of the Internet source used to communicate about medical products, the public health is best served by clear, accurate, truthful and non-misleading information about them. Read more
![]()
Scotland curbs use of transvaginal mesh amid global outcry
June 19, 2014 by Arezu Sarvestani

Scottish health regulators are looking to curb use of transvaginal mesh products while independent investigators look into claims that the medical implants hurt women.
The devices have come under increased scrutiny worldwide amid a campaign from patients and advocacy groups who warn that the devices do more harm than good. Hundreds of mesh-related lawsuits are already underway in Scotland as well as in the U.S. and Canada. The European Commission is also conducting its own investigation into the implants. Read more

