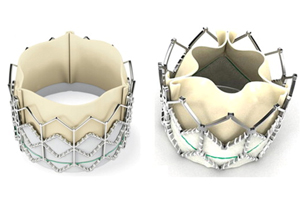
Edwards Lifesciences’ original Sapien (left) and new Sapien XT (right) valves.
Medical device maker Edwards Lifesciences (NYSE:EW) unveiled results from the 1st randomized study of its Sapien XT transcatheter aortic valve replacement system, reporting non-significant improvements in safety when compared with the original Sapien system.
The PARTNER II trial’s Cohort B was designed to demonstrate that the Sapien XT was at least non-inferior to Sapien classic in terms of safety and efficacy in treating patients too sick to undergo open-heart surgery. The study was a win in that regard, with many of the short- and long-term outcomes of the devices falling along similar lines.
At 30 days all-cause mortality for the Sapien XT was 3.5%, compared to 5.1% for the original Sapien valve, according to Edwards. Although the findings missed the mark for statistical significance, it was a victory for the Sapien line.
"These are the lowest 30-day mortality results for transcatheter aortic valve replacement in any study using rigorous clinical trial methodology," co-principal investigator Dr. Martin Leon said in prepared remarks. "For these inoperable patients, the sickest of the sick, they’re surprisingly good."
Disabling stroke rates at 30 days were also low for both groups, at 3.2% for Sapien XT and 3% for Sapien, according to Leon’s presentation.
At 1 year, all-cause mortality increased to 22.5% for Sapien XT and 23.7% for Sapien. Disabling stroke rates rose to 4.5% for Sapien XT and 4.6% for Sapien.
The PARTNER II trial is the 1st clinical study of its kind assessing the next-generation Sapien XT valve. It’s also the only study to compare Sapient XT against its predecessor, the original Sapien valve. PARTNER II was launched in part to re-examine the high rates of adverse events seen among TAVI patients in the 1st PARTNER trial, in which researchers reported that patients undergoing TAVI had a nearly doubled risk of stroke 30 days after the procedure compared with patients who underwent open-heart surgery for valve replacement. In addition, previous reviewers noted that patients who received transcatheter valve replacement had "more frequent vascular complications."
The FDA’s Circulatory System Devices advisory panel and ultimately the FDA itself decided that the device’s benefits outweigh the risks and in November 2011 handed Edwards Lifesciences the 1st U.S. premarket approval for a TAVI system.
The original Sapien is made of cow tissues and polyester supported with a stainless steel mesh frame. The device is compressed into the end of a catheter, which is slightly wider than a pencil, and inserted into the femoral artery through an incision in the leg. A physician threads the catheter to the heart where the Sapien device is released and expanded using a balloon.
The redesigned Sapien XT features material changes to the stent frame, the valve leaflet and the catheter that aim to make the device thinner and smaller. Sapien XT is a 40% lower-profile system than Sapien classic, according to the study abstract.
"With the new device, we can treat patients who have smaller vessels with less injury and less trauma, and the valve positioning is more precise," Dr. Leon said. "Any time you get below 20 French in catheter size, that’s significant for patient safety."

