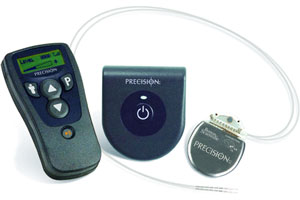
Boston Scientific (NYSE:BSX) touted CE Mark approval in the European Union for its Precision Plus spinal cord stimulator, which U.K. regulators also cleared for use during head-only MRI scanning.
The Precision Plus system, the 1st rechargeable implantable pulse generator, delivers electrical pulses that mask pain signals on their way to the brain.
Boston Scientific won FDA approval for the device in 2005 and in Europe and Canada in 2005, and more than 60,000 patients worldwide have been treated with Precision Plus thus far, according to a press release. That represents about 17% of the 350,000 patients worldwide who have received spinal cord stimulation therapy to date, according to Boston Scientific.
The new CE Mark wins allow Boston Scientific to market the system for patients with chronic pain of the trunk, and provide additional diagnostic options to patients who may need a cranial MRI.
Chronic pain is defined as continuous pain that has lasted more than 6 months, and the condition affects 1 in 5 adults in Europe, according to the company.
"As spinal cord stimulation becomes more widespread for control of severe disabling refractory pain, it is great to know that – should the need arise – head-only MRI scans can be safely performed in patients with the Precision Plus SCS system," pain management consultant Dr. Simon Thomson said in prepared remarks.
Boston Scientific Corp. (NYSE:BSX) nearly sold its neurostimulation division to Stryker Corp. (NYSE:SYK) late last year, but the Natick, Mass.-based medical device maker spiked the deal when the bid didn’t rise high enough. The company then doubled down on the pain management division with a $78 million buyout of Intelect Medical Inc., developer of the Guide deep-brain stimulation programming system.

