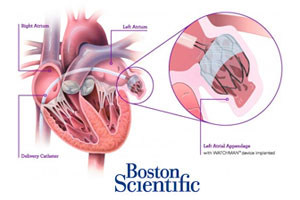
Boston Scientific (NYSE:BSX) and its Watchman anti-stroke device are next month slated for their 3rd appearance before an FDA advisory panel, the safety agency said today.
Two prior meetings of the Circulatory System Devices panel resulted in approval recommendations for the Watchman device. Despite the 1st panel’s 2009 positive vote, the FDA in March 2010 issued a "not approvable" letter for Watchman, a transcatheter implant designed to reduce the risk of stroke by closing off the heart’s left atrial appendage.
A 2nd panel voted 13-1 last year that the Watchman implant meets safety and efficacy goals. The FDA said today on its website that the circulatory devices panel is scheduled to meet Oct. 8 for another look at the pre-market approval application for the device.
Marlborough, Mass.-based Boston Scientific revealed in June that the FDA wanted to convene another panel to review the PMA application for Watchman. Today the watchdog agency said its Circulatory System Devices panel is slated to review "new clinical data and associated additional adverse events including stroke that have become available since the previous advisory committee meeting."
Back in June, Boston Scientific CFO Dan Brennan said the FDA had told the company it wanted to "look at the total body of clinical evidence up to the present time."
"We look forward to presenting the data and believe that the data and what we have support the safety and efficacy of Watchman in appropriately selected patients," Brennan told analysts at an investors conference.
The circulatory devices panel is also scheduled Oct. 9 to review cardiac allografts harvested from donor hearts, which were grandfathered in under the FDA’s 510(k) protocol but are unclassified devices, according to the agency.