Steve Kelly’s spent three decades watching new technology that originated in consumer electronics wreak havoc on well-established industries. The CEO of Myomo Inc. bore witness as new technologies redefined the home computing market in the 1980s, the telecom space in the 1990s and the changes voice-over-IP technology brought to telephony in the last decade.
Kelly sees a similar wave heading for the shores of the medical device industry. MassDevice spoke with him to hear his take on where the sector is headed and why.
MassDevice: Can you give us a quick primer on the origins and history of Myomo?
Steve Kelly: This technology comes out of the lab of Prof. Woodie Flowers. Woodie was one of the leading professors in mechanical engineering and robotics at MIT. He developed the Boston Arm, he was the first host of the American Scientific show that Alan Alda does, co-founded FIRST Robotics with Dean Kamen.
A couple of his students came up with the idea. There are prosthetics for amputatees, but for people who have some sort of muscular impairment, why isn’t there something to help them? So it began as a research project. They won the MIT 50k [business plan contest] back a few years ago it got spun out as a company and today the company has an arm product on the market, the e100, that is cleared for treating people who have a stroke.
MassDevice: What’s your background? How did you come to a career in medical devices, and what’s the appeal of this industry for you personally?
SK: I’ve worked in technology for about 30 years now and I keep seeing the same cycle repeated in different industries. Basically, low-cost computer and electronics components migrate into a new market segment and then completely redefine that industry. In the 80s I worked for Digital Equipment Corp. when the PC came along. And then in the 90s I worked in the networks and communications space when those components kind of redefined communications, and along came the Internet. Then I worked in a similar vein in the telephony space in what brought about voice-over-IP.

I started to volunteer time back a few years ago at MIT to work with late-stage research projects like this one and basically saw that the same thing is about to happen to the medical device business. I’m particularly excited about it happening in the space known as the orthotics and prosthetics market.
Medical devices will increasingly be derived from what are basically consumer electronic components. That will drive down their cost. The smaller and smarter components — things like batteries, processors, storage and motors — will lead to an entirely new class of products. If you look at Myomo’s product as an example, there are products that are kind of rehab-hospital, gym-sized equipment that sell for tens and hundreds of thousands of dollars. The Myomo product is basically redefining that with a shift to more personal technology. We’re delivering similar capabilities for an order of magnitude less cost, and we’re developing portable products that allow them to be used in an in-patient setting, to help somebody get out of bed, all the way into a home setting where a home health visiting nurse therapist goes in and helps somebody get acclimated when they’re discharged home.
This is the best, most fulfilling job I think I’ve ever had. There are all sorts of medical devices; a lot of them are used in surgery and it’s gratifying. But when you watch somebody put this on and for the first time in a week, month or upwards of 20 years move their arm again, oh my gosh it’s like an electric current going through you. One of the people I learned a lot from when I went to do my first start-up, I asked him for advice. First of all, he said, find a big problem to solve. With three million people who’ve survived a stroke but are impaired, I feel like that’s a pretty worthy problem to go after solving.
MassDevice: Myomo had some early success, landing FDA clearance in July 2007 for the e100 NeuroRobotic System, but ran into some financial difficulties in 2008 that forced some layoffs. Have things improved since then?
SK: Things are going pretty well. I’m pretty pleased. This is really a new type of technology, so we’ve been trying to take this in kind of a measured pace to really get things right. Right now we’re deployed pretty well in the Northeast. We’ve begun to do some expansion beyond that, but we haven’t announced any of those clinical partners yet. We’ve begun to put the prod into the home by way of home health agencies. For us that’s a really good structured next step from being in the clinic because the therapist is right there working with the person in the home. This is something we’ve seen put on people a week to 20 years post-stroke and within a couple of minutes they’re moving their arm again in a way that they were not able to before.
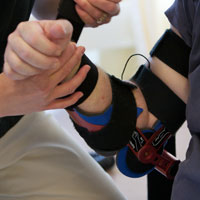
The thing that it seems to really leverage is the concept of neuroplasticity. Two things that really seem to hit that sweet spot are the portability of it and the fact that it’s really the person controlling the device, rather than the device controlling the person. That means you can put it on somebody, whether you’re in the rehab or the home, and they can use it to focus on functional tasks, which is what the literature says is the way to maximize promoting neuroplasticity. If you just put somebody’s arm in a machine and have the machine move it, that doesn’t seem to do anything to interest your brain. When you put this on someone and they start working on flipping the light switches and getting a cup to their mouth or moving a laundry basket, well, that seems to really have an impact.
The second aspect of it that’s really essential is repetition. Somtimes repetition can be pretty darn boring. The fact that you can put this on and do things that are meaningful to you, well, all of a sudden that kind of repetition can be pretty gratifying. That’s a lot different from being sent home and being able to move your arm maybe 10 degrees and be told, "Every day I want you to sit there and move that arm back and forth as much as you can." That’s just demoralizing, if you don’t feel like it’s making a difference and you have to do everything one-handed. This device has gotten people to the point that they’re able to do things without the device that initially they were only able to do with it.
MassDevice: You’ve had success raising capital through equity offerings (most recently raising about $617,000), to the tune of a reported $5 million. How much have you raised altogether?
SK: We don’t disclose, SEC filings aside, any of the financing numbers publicly. I can tell you that we’re well-financed right now and if you look at the size of the market potential for this I wouldn’t be surprised to be doing additional rounds of financing.
MassDevice: What was the outcome of the 20-patient clinical trial in Cincinnati?
SK: That study is still going on. It was combined with some underwriting by the American Heart Assn. that adds kinematics to the study, kinematic analysis. We also doubled the size of the study to 40 patients. It’s a two-year study that we expect to finish next year. The preliminary results have been really positive, meaning that we’ve seen improvement in all of the subjects so far that have completed the protocol. We’re pretty excited about this. If I had to summarize who it’s really beneficial for, there are three million moderately to severely injured stroke survivors who have difficulty moving their arm. This seems to be a really good solution for that population.

The vision that we have for this is to have an entire family of Myomo products for not just the arm, but the hand, the ankle, the knee, et cetera. You should be able to wheel somebody in and fit them up with the Myomo parts that they need and have them be able to walk out relatively unassisted, depending on their condition. They may have a degenerative condition, where over time they’ll need more, or because of whatever treatment they’re getting, they may be able to wean themselves off of Myomo — or, in the case of stroke survivors, the product itself may have a role in people’s recovery, being able to wean themselves off of the product.
One of the things that most people do not know is that about a third of stroke survivors are under the age of 65. So you get a lot of young mothers; it’s not uncommon for someone in active combat, when theyre shot, to shoot a clot and have a stroke. It’s the single leading cause of permanent disability, so it just touches almost everyone’s life at some point.

