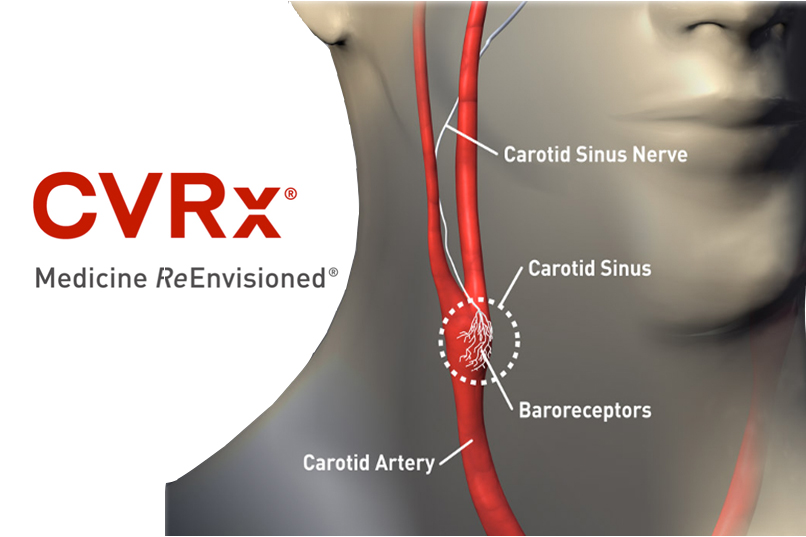
CVRx said today that it won CE Mark approval in the European Union for its Barostim Neo device to treat heart failure patients using vagus nerve stimulation.
CVRx president & CEO Nadim Yared told MassDevice.com today that the device is designed to influence both the sympathetic and parasympathetic systems, reducing sympathetic activity and increasing parasympathetic activity.
"It’s actually pretty straightforward. Heart failure as a disease is most often due to the fact that the sympathetic signal from the brain to the organ is elevated and the vagus tone is depressed. It’s the same with hypertension, basically. We have a unique platform where we are not targeting either the sympathetic system alone or the parasympathetic system alone," Yared told us.
Last week, he noted, CVRx added a $15 million loan from Silicon Valley Bank to the $251 million it’s raised so far. CVRx is negotiating with the FDA on the parameters for clinical trials in the U.S. ahead of pre-market approval bids for both the heart failure indication and the hypertension indication, Yared said (the Minneapolis-based company won CE Mark approval for the treatment of hypertension in August 2011).
The company is also investigating the Barostim treatment for acute kidney disease with strategic partner DaVita (NYSE:DVA), he added. But it’s the heart failure indication that holds the most promise, given that some ⅔ of heart failure patients aren’t eligible for cardiac rhythm therapy (and that a portion of patients who are eligible for CRT don’t respond), Yared explained.
"The sum of those 2 make it a very large market for our therapy in Europe," he said. "Once the optimal medical treatment is given to those [ineligible for CRM] patients, if they stay in Class III heart failure, they are still at a very elevated risk of death or morbidity."