The battle between surgical devices maker Intuitive Surgical (NSDQ:ISRG) and analysts at Citron Group continues to heat up, with harsh words from one and a stern defense from the other.
Citron Group today issued an open letter to Intuitive Surgical, accusing the company of misusing FDA data to falsely paint a picture of declining adverse events associated with the da Vinci surgical robot.
Both companies have ceded that the FDA’s MAUDE adverse event reporting system is flawed at best, and the FDA itself has warned against using the database to attempt to infer a device’s safety profile, but a recent surge of patient injury and death reports associated with Intuitive’s flagship da Vinci system has raised a few eyebrows.
""In the 1st 8 months of 2013, 2332 Adverse Event records were posted – compare to 4603 records posted in the entire 12 year period since the 1st Adverse Event tracking for da Vinci appeared in MAUDE in 2000," Citron wrote. "It is the opinion of Citron that the only reason there is not a national outcry is because the da Vinci robot has yet to kill or injure ‘the right person’ – like the next of kin of a congress member or a celebrity."
"What is important to note is that MAUDE is not meant to compare rates of da Vinci versus (other devices) laparoscopic instruments or open surgical tools," according to a statement Intuitive sent to MassDevice.com today. "This guidance from the FDA is the reason why we strongly encourage surgeons and patients to look at peer-reviewed clinical data when making an assessment about the safety and efficacy of robotic assisted surgery with da Vinci."
Citron analysts have been stark in their criticisms of Intuitive, saying the company is allowing under-trained surgeons to perform complicated procedures while ignoring signs that patients are meeting with harm. Intuitive responded by standing behind its data, citing FDA guidance on the agency’s adverse event reporting database and pointing to clinical data as evidence of the da Vinci system’s ever-improving safety profile.
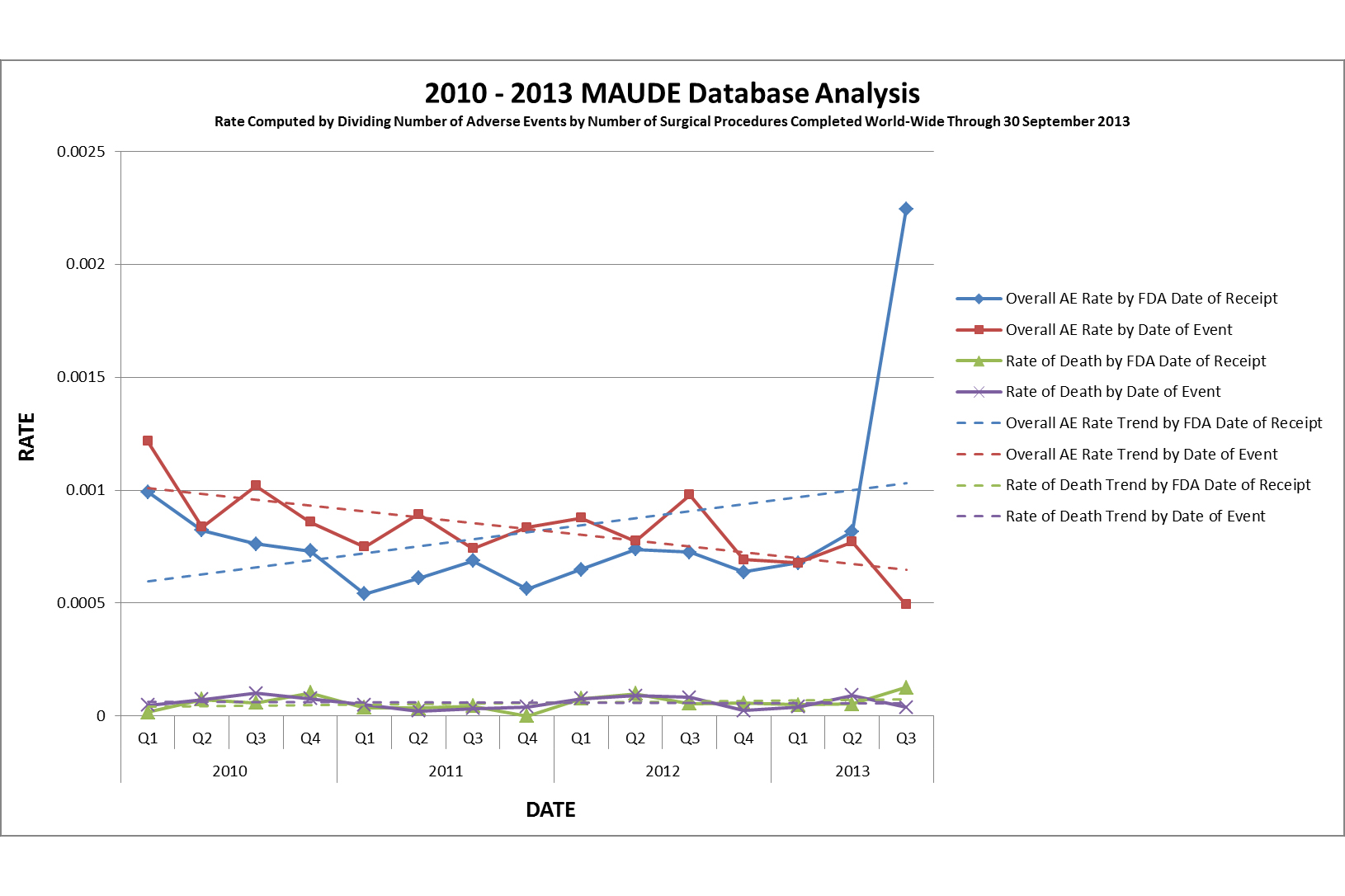
Citron took issue in particular with Intuitive’s use of a graph that the company said clarifies the difference between the dates the adverse events were reported to the MAUDE database and the dates the incidents actually occurred. Some of the reports were filed months or even years after they took place.
The companies have been at it for some time, since Citron in January 2013 published all of the FDA’s da Vinci adverse event filings, comprising 12 years’ worth of reporting. Earlier this month Citron released another report, accusing Intuitive Surgical of ignoring a surge of new incidents that have cropped up in the FDA’s system.
Citron analysts chided the company as well as other analysts for underestimating the concerns that have dragged down ISRG shares by about a third since this time last year. Intuitive Surgical in recent months has been the target of an FDA probe into surgical complication rates, an American Congress of Obstetricians & Gynecologists statement questioning the benefits of da Vinci surgery, a high-profile patient injury lawsuit painting the company as an aggressor, a surgical instrument crack warning and a shareholder class action lawsuit against the company and its leadership.
Intuitive Surgical spokeswoman Angela Wonson denied that there was a true increase in da Vinci incidents, saying that Citron examined the dates the events were reported, rather than when they actually occurred. The company has maintained that its analysis shows a decrease in adverse events when looking at global rates of surgery compared with FDA reports, but Citron took issue with that as well.
"As you well know, it is extremely unlikely that FDA Adverse Event Reports on surgeries performed outside the United States will ever be filed," according to Citron’s open letter. "Nevertheless, you choose as a denominator ‘completed worldwide surgical procedures’."
Intuitive responded that the FDA accepts and contains adverse events from outside the U.S., and "therefore we use global numbers for the denominator."