
From pulsed field ablation to diabetes, surgical robotics, and plenty more, a number of medical device launches sit on the horizon in 2024.
Pulsed field ablation news took over at the end of 2023 and the start of 2024, with Medtronic and Boston Scientific garnering significant regulatory approvals. Surgical robotics remains a hot space, and some of the orthopedic giants are continuing to make waves there. Plus, heart valves, neurostimulation components, drug-coated balloons, and more are likely to hit the market sometime this year.
Here’s a list of 10 significant medical device launches we could see in 2024.
 Part of the Farapulse pulsed-field ablation system, the Farawave single-shot catheter is designed to create durable and circumferential lesions. [Image courtesy of Boston Scientific]
Part of the Farapulse pulsed-field ablation system, the Farawave single-shot catheter is designed to create durable and circumferential lesions. [Image courtesy of Boston Scientific]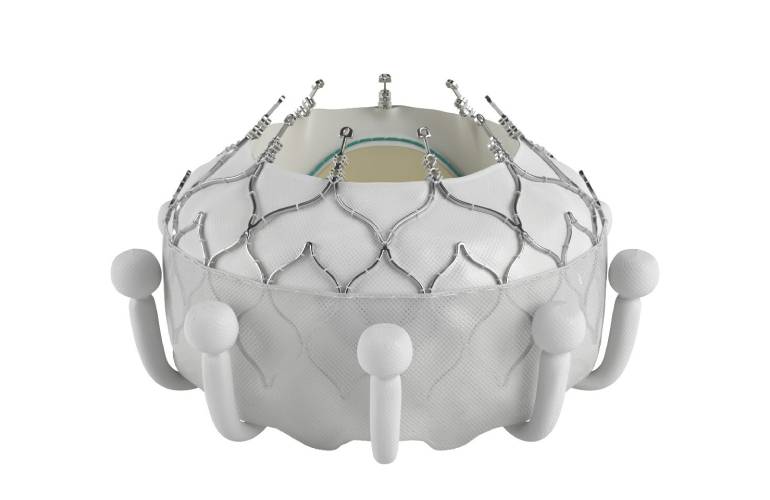 The Evoque tricuspid valve replacement system. [Image courtesy of Edwards Lifesciences]
The Evoque tricuspid valve replacement system. [Image courtesy of Edwards Lifesciences]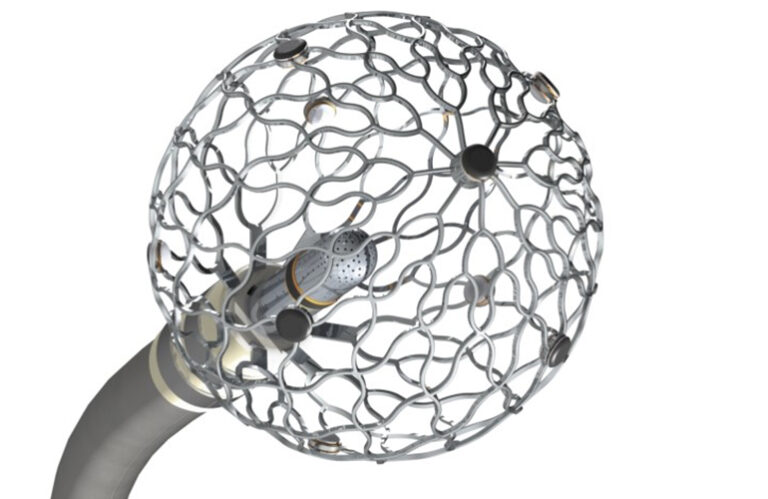 Affera’s Sphere-9 mapping and ablation catheter [Photo courtesy of Affera]
Affera’s Sphere-9 mapping and ablation catheter [Photo courtesy of Affera]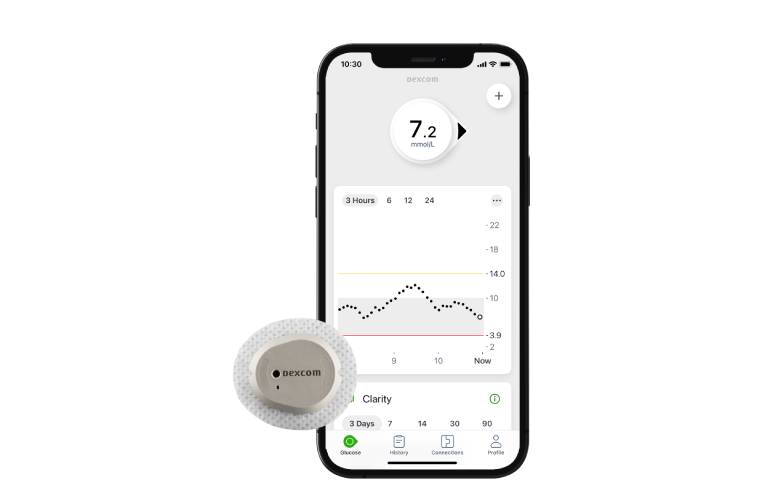 The G7 CGM. [Image courtesy of Dexcom]
The G7 CGM. [Image courtesy of Dexcom] The Mako robotic-assisted surgery system. [Image courtesy of Stryker]
The Mako robotic-assisted surgery system. [Image courtesy of Stryker]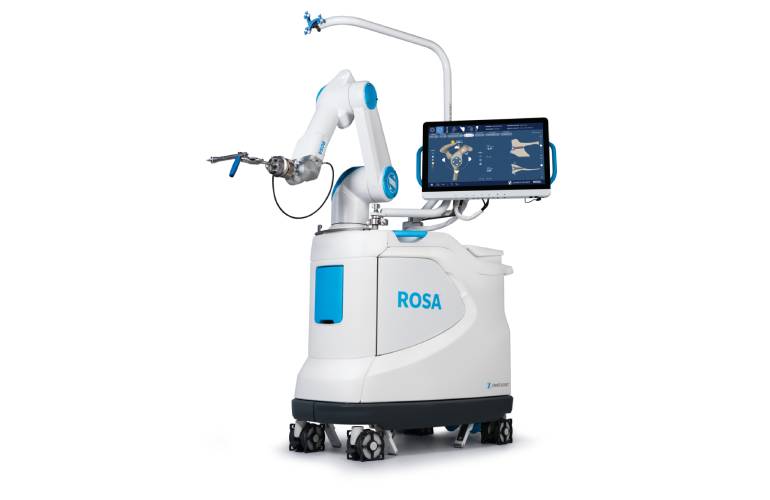 The Rosa Shoulder surgical robot system. [Image courtesy of Zimmer Biomet]
The Rosa Shoulder surgical robot system. [Image courtesy of Zimmer Biomet] The Agent drug-coated balloon. [Image from Boston Scientific]
The Agent drug-coated balloon. [Image from Boston Scientific]Click the ‘+’ symbol to expand.
Device: Farapulse
Launch timeframe: Estimated to launch in the second half of 2024
Farapulse’s nonthermal treatment uses electric fields to selectively ablate heart tissue. Boston Scientific bought Farapulse for nearly $300 million in 2021. The system delivers pulsed field energy with the Farawave ablation catheter, Farastar ablation generator and Faradrive steerable sheath. Boston Scientific’s VersaCross Connect access solution complements the sheath to provide safe and effective left heart access. The Farawave catheter treats a range of pulmonary vein anatomies using an over-the-wire catheter with a variable basket and flower shapes. This allows for the device to adapt to individual patient anatomies. Boston Scientific won FDA approval for Farapulse in January.
Device: Evoque Tricuspid
Launch timeframe: Estimated mid-2024
Edwards Lifesciences designed Evoque for tricuspid regurgitation. It won FDA approval in February, making it the first transcatheter therapy to receive an FDA nod to treat tricuspid regurgitation. Evoque features a nitinol self-expanding frame, intra-annular skirt and tissue leaflets made from bovine pericardial tissue. Edwards intends to offer the valve in three sizes, all delivered through the same low-profile transfemoral 28F system.
Device: Affera PFA
Launch timeframe: Estimated second half of fiscal 2024/First half of 2025
Medtronic first made waves in the PFA space with its 2022 $1 billion acquisition of Affera and has since made significant progress in that space. Affera technology makes up a key component of Medtronic’s PulseSelect PFA system, which became the first such system FDA-cleared for treating AFib late last year. It also provides mapping and ablation within the Sphere-360 investigational PFA catheter technology.
In January, CEO Geoff Martha reported “good progress” in bringing the Affera Sphere-9 PFA mapping and ablation catheter to market this year. The Affera system received CE mark more than a year ago.
Device: Non-insulin designed CGM
Launch timeframe: Likely second half of 2024
Dexcom designed its Stelo CGM with a 15-day wear and a software experience tailored specifically for non-insulin users. At the company’s June Investor Day event, it revealed its plans to bring the new product to market in the U.S. in 2024. According to Dexcom, the target population includes approximately 70% of Americans with diabetes. The device won’t “bother” users with the alerts and alarms geared toward insulin users, Dexcom CEO Kevin Sayer told Drug Delivery Business News shortly after the company’s submission to the FDA. Rather, it just offers the insights that users desire. Stelo exists on the current G7 platform, so Sayer said manufacturing is already in place on the current lines the company has. Notably, rather than the initially touted cash-pay option, Stelo received FDA clearance as an over-the-counter offering in March.
Device: 15-day G7
Launch timeframe: Second half of 2024 or later
Dexcom launched its next-generation G7 CGM more than a year ago. That device — the G7 — offers a 10-day wear time, but the company looks set for an upgrade from 10 days to 15 days. The extended-wear version of the leading CGM offering could be on the market some time this year.
See the full list of diabetes devices set to launch in 2024.
Device: Mako spine/shoulder
Launch timeframe: Spine: Mid-2024/Shoulder: Second half of 2024
Early last year, Stryker suggested that its Mako surgical robotic platform would have significant application expansions in 2024. It said it expected a Mako Spine launch in the second half of 2024, with Mako Shoulder out by the end of 2024. Mako Spine is an application that combines with the company’s new bone-cutting product. Then, Mako Shoulder will feature a robotic application that adds incremental precision. Stryker continues to expand Mako, unveling a new medium to extend a surgeon’s Mako SmartRobotics experience in and beyond the operating room earlier this year. The company’s myMako app for Apple Vision Pro and iPhone allows surgeons to visualize and review patients’ Mako surgical plans anytime, anywhere.
Device: ROSA Shoulder
Launch timeframe: Mid-2024
Zimmer Biomet won FDA clearance for its ROSA Shoulder robotic-assisted surgery system in February. The company designed ROSA Shoulder to allow surgeons to execute total shoulder replacement using anatomic or reverse techniques. It enables precise placement for improved outcomes. According to Zimmer Biomet, it’s one of the only systems that can reproduce humeral head resectioning and ease the insertion of instruments into incisions by requiring no pin in the center of the glenoid during procedures.
Device: Artix
Launch timeframe: Estimated 2024
The Artix MT thrombectomy device is indicated for the non-surgical removal of emboli and thrombi from blood vessels. It can also be used for injection, infusion or aspiration of contrast media and other fluids into and from a blood vessel. The company designed it for use in the peripheral vasculature. Artix combines mechanical and aspiration thrombectomy and proximal flow arrest to prevent distal embolization. The sheath has four times the flow rate versus existing arterial catheters, according to the company.
Device: Inspire V
Launch timeframe: Limited market release: Estimated second half of 2024; Full market release: Estimated first half of 2025
Implantable pulse generators (IPGs) deliver low-intensity electrical impulses to nerve structures, allowing neurostimulation technologies to deliver therapeutic value. A next-generation IPG with a removed sensing lead could enhance the therapy delivered by Inspire Medical’s minimally invasive implantable products for treating obstructive sleep apnea. The devices offer an alternative to the traditional CPAP machines made by the likes of Philips and ResMed. Inspire Medical’s reputation has grown over the past several years as Philips dealt with a massive CPAP recall and people moved away from that method of treatment.
Device: Agent
Launch timeframe: Estimated second half of 2024
The FDA approved Boston Scientific’s Agent drug coated balloon in March. The DCB won approval for treating coronary in-stent restenosis (ISR) in patients with coronary artery disease. ISR occurs when plaque or scar tissue obstructs or narrows a stented vessel. Boston Scientific said it plans to launch the device in the coming months. Agent serves as an alternative to traditional therapies like balloon angioplasty, additional layers of stenting or radiation. The paclitaxel-coated balloon transfers a therapeutic dose of the drug to the vessel wall, helping to prevent ISR reoccurrence.
Device: Precision7
Launch timeframe: Estimated 2024
Alcon is set to launch its Precision7 reusable lens for seven-day wear. The lens is intended to correct vision by focusing light on the back of the eye, allowing the person wearing it to see a clear image. The company also designed the lens to treat near-sightedness (myopia), far-sightedness (hyperopia) and blurred or distorted vision due to an irregular curvature of the cornea, or front surface of the eye (astigmatism). These lenses also correct presbyopia that leads to a gradual inability to see things clearly close-up.

