FDA withdraws older guidance documents
The FDA is withdrawing 47 draft guidance documents published before the end of 2013 that it never finalized or acted on.
Agency officials posted the announcement in the Federal Register, explaining that the move is designed to boost efficiency and transparency regarding its development of guidance documents.
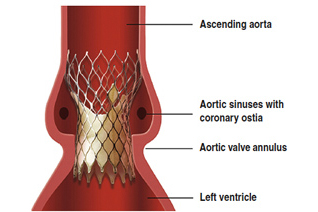
Sorin hopes Perceval valve will rescue flat results
Diabetes: J&J plans mobile app trial for OneTouch Verio Flex device
Medtronic launches new spine devices
Oculus Rift heads to the neurosurgery suite
Axonics generates another $17m
Axonics Modulation Technologies said it raised nearly $17.3 million in an equity financing as it develops next-generation neuromodulation technology to treat various medical conditions.
The California company disclosed the funding in a recent regulatory filing that does not name specific investors.
TAVI: U.K. study finds 60% survival rate at 3 years
Profound Medical goes public in reverse merger
Profound Medical yesterday said it’s slated to hit the public markets in Canada after a reverse merger, also revealing a $24 million private placement to fuel clinical trial work.